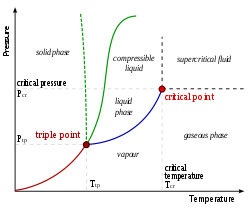
 As these gas phase molecules move randomly about, they will occasionally collide with the surface of the condensed phase, and in some cases, these collisions will result in the molecules re-entering the condensed phase. When a liquid vaporizes in a closed container, gas molecules cannot escape. In this module, the essential aspects of phase transitions are explored. These changes of state are essential aspects of our earth’s water cycle as well as many other natural phenomena and technological processes of central importance to our lives. 
As one example of global significance, consider the evaporation, condensation, freezing, and melting of water. We witness and utilize changes of physical state, or phase transitions, in a great number of ways. Describe the processes represented by typical heating and cooling curves, and compute heat flows and enthalpy changes accompanying these processes.Explain the relation between phase transition temperatures and intermolecular attractive forces. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
